 What is your body’s largest internal organ that filters and extracts toxins from your blood and can even regenerate on its own?
What is your body’s largest internal organ that filters and extracts toxins from your blood and can even regenerate on its own?
The liver!
Although the liver may not be discussed as frequently as the brain or heart, the liver is an incredibly critical part of everyday life and plays a role in metabolism, digestion, immunity, and overall keeping the body healthy. On April 19th we celebrate World Liver Day and honor everything it does for our bodies. Just some of the liver’s major roles include the production of bile (which helps break down fats during digestion), detoxification of the blood supply, and storage of essential vitamins (Kalra et al., 2023). The complex involvement of the liver in all these functions means that any injury or damage can cause a cascade of negative consequences. That being said, there are many useful preventative and curative measures that people can utilize to heal the liver. In this post, we gained insight from liver experts, clinicians, and people with lived experience to highlight the good, the bad, and the healthy for all things liver!
Liver Disease
So, what exactly causes someone to get a liver disease?
It all boils down to tissue damage which can be caused by viral infections, excessive alcohol consumption and fat intake, autoimmune disease, and more.
Viral infections
Of course, one of the most common diseases that affects the liver is hepatitis in all its forms. Hepatitis translates to inflammation of the liver, which can be caused by viral infections, excessive alcohol consumption, or autoimmune diseases. Regarding viral infections there are five different hepatitis viruses (A, B, C, D, and E) that can damage the liver. Dr. Su Wang, Senior Advisor for Global Health for the Hepatitis B Foundation and health care provider, explains the mechanism for viral liver damage.
“The virus resides and replicates in the liver and over time can cause chronic inflammation which can lead to fibrosis [mild scarring] and even progress to cirrhosis [severe scarring] and liver cancer.”
Dr. Robert Gish, hepatologist and Medical Director for the Hepatitis B Foundation, highlights how the different viruses affect the liver.
“Hepatitis A only causes acute infections, meaning the body can recover eventually, whereas hepatitis B, C, D, and E can cause chronic infections that are more likely to lead to long-term damage if not managed.”
The immune response from liver cells while trying to protect against hepatitis virus can lead to long term inflammation and damage of cells. This causes fibrous connective tissue to develop within the organ as a response to injury and is often one of the first steps towards liver disease.
Alcohol and Diet
Excessive fibrosis development and scarring of the liver can lead to a disease called cirrhosis which can also come from excessive alcohol consumption. Since your liver filters toxins out of the blood, the process of filtering alcohol out of the blood causes injury and cell death. Read more about alcohol’s effect on the liver in this blog post.
Another common method of liver damage is through dietary fat deposits that accumulate in the liver referred to as steatosis. This can lead to diseases known as metabolic-associated steatotic liver disease (MASLD), alcoholic steatotic liver disease (MetALD), and metabolic-associated steatohepatitis (MASH). Wayne Eskridge, CEO of the Fatty Liver Foundation explains more about these common liver diseases and how to recognize their presence.
“Metabolic-associated steatotic liver disease (MASLD) is the new term for nonalcoholic fatty liver disease (NAFLD). It is characterized by excess fat in the liver not caused by excessive alcohol consumption.
Alcoholic-associated steatotic liver disease (MetALD) is the new term for what was previously known as alcoholic fatty liver disease (AFLD). It is caused by heavy alcohol use.
The stages of MASLD are as follows:
- Simple steatosis – Fat accumulation in the liver, but no inflammation or damage to liver cells.
- Metabolic associated steatohepatitis (MASH) – Inflammation and damage to liver cells, along with fat accumulation. MASH can lead to cirrhosis and liver failure.
- Cirrhosis – Scarring of the liver due to chronic inflammation and damage. Cirrhosis can lead to liver failure and other complications, such as portal hypertension (high blood pressure in the portal vein) and ascites (fluid buildup in the abdomen).”
MASLD and MetALD are becoming more common in the general population, particularly MASLD, likely becasue of the increasing prevalence of obesity, diabetes due to a sedentary lifestyle, and eating diets that are high in processed foods, sugary drinks, and unhealthy fats that can promote liver fat accumulation.
Less Common Conditions
In addition to the more common liver diseases, there are some lesser-known conditions that are genetic and autoimmune. Wayne discusses four that he identifies as important for people to know.
Primary Biliary Cholangitis (PBC) – A chronic autoimmune disease that affects the bile ducts in the liver, leading to inflammation and damage
Autoimmune hepatitis – This is a chronic inflammatory condition where the immune system attacks the liver, leading to liver inflammation and damage
Hemochromatosis – This is a genetic disorder where the body absorbs too much iron, leading to iron overload in the liver and other organs (Read blog post about hemochromatosis here)
Wilson’s Disease – A rare inherited disorder that causes copper to accumulate in the liver, brain, and other vital organs
All liver diseases mentioned in this section can cause excessive stress and changes to the liver tissue, which can lead to liver cancer development. It is important to note though, that fibrosis is reversible and there are things you can do to maintain your liver health to avoid advanced disease.
Healing the Liver
The liver can be negatively affected in many ways, but the good news is that the liver is an incredibly resilient organ and there are many ways to heal the liver.
Treatment
Clinicians and patients agree, the most important step to healing the liver from a viral infection is treatment. Dr. Gish states that for hepatitis C, “cure with direct-acting antivirals is the first step” and hepatitis C lived experience expert, Courtney Downs, agrees:
“To heal your liver while living with hepatitis C, prompt treatment is key. Early treatment enhances a person’s overall health and leads to numerous benefits such as: improved quality of life, decreased chances of exposing others, regression of liver fibrosis and a reduction in the risk of liver failure and liver cancer – hepatocellular carcinoma (HCC)”
Although there is not a cure for hepatitis B like there is for hepatitis C, there are many available and accessible treatments for hepatitis B that can stop viral replication and liver damage. Learn more about available hepatitis B treatment options here.
Dr. Wang highlights additional management considerations including asking for lab tests to measure “viral load and liver enzymes at least once a year if not more often” and recommends having discussions with your healthcare provider. “You want to ask about liver cancer screening as well and have an Alpha Fetoprotein (AFP) test and ultrasound ordered”. These steps will help to monitor the liver status and prevent progression of liver damage to irreversible stages of cirrhosis and cancer.
In terms of fatty liver diseases, there may be medications that can be prescribed to reduce liver fat or improve liver function and in severe cases, a liver transplant may be necessary.
Lifestyle
There are a few lifestyle changes that all people can make to reduce the negative effects of liver disease and overall improve liver health regardless of disease status, recommended by Mr. Eskridge and Dr. Gish:
- Limiting Alcohol Consumption – Alcohol use at any level can put greater stress on the liver. Rethink the drink!
- Dietary Changes – Focus on fruits, vegetables, whole grains, unsaturated fats, and lean protein. Limit processed foods, sugary drinks, and saturated or trans fats.
- Increasing Exercise – Aim for at least 150 minutes of moderate-intensity exercise per week.
Courtney provides her insight as someone with experience living with a liver disease.
“The liver has the unique capacity to regenerate once no more virus is detected and if no new damage occurs. Optimal nutrition and alcohol avoidance are vital. If abstinence isn’t feasible, reducing alcohol and consuming lean protein can aid liver repair. Also, getting vaccinated against hepatitis A and B is very important to protect your liver.”
There are safe and effective vaccines to prevent hepatitis A and B infections. This is an easy step to protect your liver from future damage and prevent simultaneous liver disease while the liver is in a weakened state. Learn more about the hepatitis B vaccine here.
Field experts interviewed for this post highlighted their favorite fact about the liver and why World Liver Day is important to them:

Dr. Robert Gish
“You cannot live without your liver.”
“Raising awareness will change human behavior and lead to better liver health.”

Dr. Su Wang
“The liver can regenerate, and fibrosis can be reversible. It is such an important organ and makes almost all the important proteins in our body but also detoxifies.”
“The liver is an organ that doesn’t get enough attention given its importance and because of that, liver diseases are often neglected. Thus, this day is even more important to increase awareness and promote liver health.”

Wayne Eskridge
“Education is the only real answer to stemming the tide of this disease (MASLD/MetALD/MASH). Drug therapy is not sustainable on a population basis so people must learn to develop better habits.”
Hepatitis C lived experience expert, Courtney, expressed advice for anyone living with hepatitis C or other liver diseases:

Courtney Downs
“You are not alone. There is a whole community of individuals that have cured or cleared their hepatitis C. There are clinicians, support staff, advocates, coalitions, and organizations that consist of some of the most amazing people I have ever met personally and/or professionally. They work tirelessly advocating to remove barriers and challenge the stigma that surrounds viral hepatitis. They don’t judge and they truly embrace radical love and acceptance. There is a cure and there are people who will help you get that cure no matter what your circumstances are. So, my advice would be to get tested, get treated, and get vaccinated.”
Courtney said it best. Celebrate World Liver Day on April 19th and every day.
Get tested/treated/vaccinated today!
References:
Kalra A, Yetiskul E, Wehrle CJ, et al. Physiology, Liver. [Updated 2023 May 1]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK535438/











 Amina asked her doctor how she could get rid of this virus. Her doctor explained to her that there is no cure for hepatitis B, but antiviral treatment options do exist. While she may not be able to completely get rid of the virus, she can help protect herself from serious conditions such as hep B related cirrhosis or liver cancer with treatment. Amina’s doctor encouraged her to get treatment to prevent any serious problems from occurring. He also mentioned that treatment for hepatitis B is safe and effective. This did not make any sense to Amina. She thought to herself that if a treatment wouldn’t cure her of the disease, then there is no point in taking it. She felt healthy and did not show any symptoms. After the doctor suggested treatment options, she said that she will wait for the cure.
Amina asked her doctor how she could get rid of this virus. Her doctor explained to her that there is no cure for hepatitis B, but antiviral treatment options do exist. While she may not be able to completely get rid of the virus, she can help protect herself from serious conditions such as hep B related cirrhosis or liver cancer with treatment. Amina’s doctor encouraged her to get treatment to prevent any serious problems from occurring. He also mentioned that treatment for hepatitis B is safe and effective. This did not make any sense to Amina. She thought to herself that if a treatment wouldn’t cure her of the disease, then there is no point in taking it. She felt healthy and did not show any symptoms. After the doctor suggested treatment options, she said that she will wait for the cure. After moving to the U.S., Amina had gotten busy with school and work and did not follow up with her primary care doctor for years. Amina experienced stomach pains from time to time but they often went away on their own. On one occasion, her stomach pain worsened. She had to take a few days off from work to get better using home remedies, but they didn’t help. Finally, she went to the doctor’s office to learn more. She discovered that she had liver cancer. Her doctor referred her to a hepatologist (a liver specialist) for further treatment.
After moving to the U.S., Amina had gotten busy with school and work and did not follow up with her primary care doctor for years. Amina experienced stomach pains from time to time but they often went away on their own. On one occasion, her stomach pain worsened. She had to take a few days off from work to get better using home remedies, but they didn’t help. Finally, she went to the doctor’s office to learn more. She discovered that she had liver cancer. Her doctor referred her to a hepatologist (a liver specialist) for further treatment.  The hepatologist explained to Amina that hepatitis B can lead to liver cancer without monitoring and treatment. Even though a cure is not available, treatment options do exist, and they help in slowing and preventing serious liver disease, liver damage or liver cancer. If Amina had started antiviral treatment on time, she could have saved her liver. The doctor recommended chemotherapy for Amina to treat the cancer. Not only did her medical bills go up but Amina felt physically and mentally exhausted by the procedures. She advocates for everyone living with hepatitis B to get treatment if they need it and not wait for the cure. She also participates in advocacy efforts to make treatment options more affordable for people living with hepatitis B.
The hepatologist explained to Amina that hepatitis B can lead to liver cancer without monitoring and treatment. Even though a cure is not available, treatment options do exist, and they help in slowing and preventing serious liver disease, liver damage or liver cancer. If Amina had started antiviral treatment on time, she could have saved her liver. The doctor recommended chemotherapy for Amina to treat the cancer. Not only did her medical bills go up but Amina felt physically and mentally exhausted by the procedures. She advocates for everyone living with hepatitis B to get treatment if they need it and not wait for the cure. She also participates in advocacy efforts to make treatment options more affordable for people living with hepatitis B.







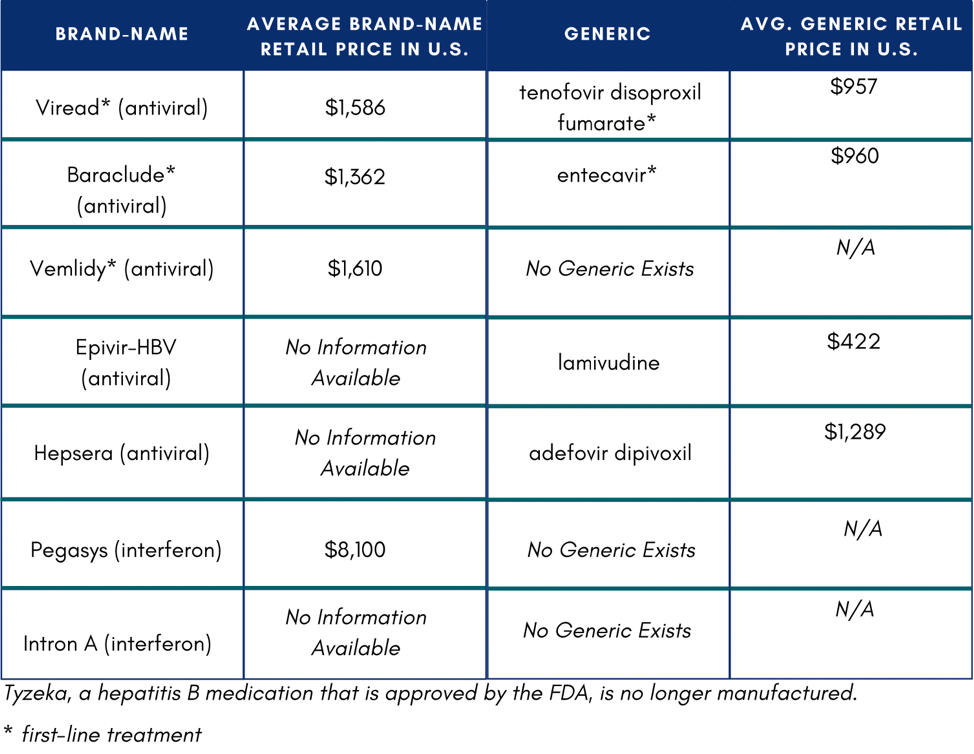 In the U.S., if someone does not have insurance or know how to access Medicaid or Medicare, they might not be able to afford medication. If they were to pay out of pocket, medication would total $11,484 on the low end of costs.4 One study reported that low household income and publicly funded health insurance were negatively associated with willingness to accept hepatitis B treatment.5
In the U.S., if someone does not have insurance or know how to access Medicaid or Medicare, they might not be able to afford medication. If they were to pay out of pocket, medication would total $11,484 on the low end of costs.4 One study reported that low household income and publicly funded health insurance were negatively associated with willingness to accept hepatitis B treatment.5