
By Sierra Pellechio, BS, CHES, Hepatitis Delta Connect Program Manager
Historically, testing for hepatitis delta has been difficult to access and often not commercially available. With the rise in awareness about hepatitis B and delta coinfection, more tests are beginning to be offered by multiple labs for clinicians in the United States looking to test their patients. Because hepatitis delta can only infect people who also have hepatitis B, the Hepatitis B Foundation’s medical director and leading hepatologist Dr. Robert Gish recommends testing all hepatitis B patients for hepatitis delta. “Screening all hepatitis B patients will allow a better understanding of hepatitis delta prevalence and its impact on outcomes and will identify patients who can be offered treatment within or outside clinical trials.”
The first step in diagnosing an infection is the HDV antibody total (anti-HDV) test. Patients who have recovered from or are currently infected will be positive for the anti-HDV and will present high titers in later stages of acute infection and persist in cases of chronic infection. If the HDV antibody total test is positive, it should be followed by the HDV RNA (PCR) test to confirm an active infection. If this test is negative, a current infection is unlikely.
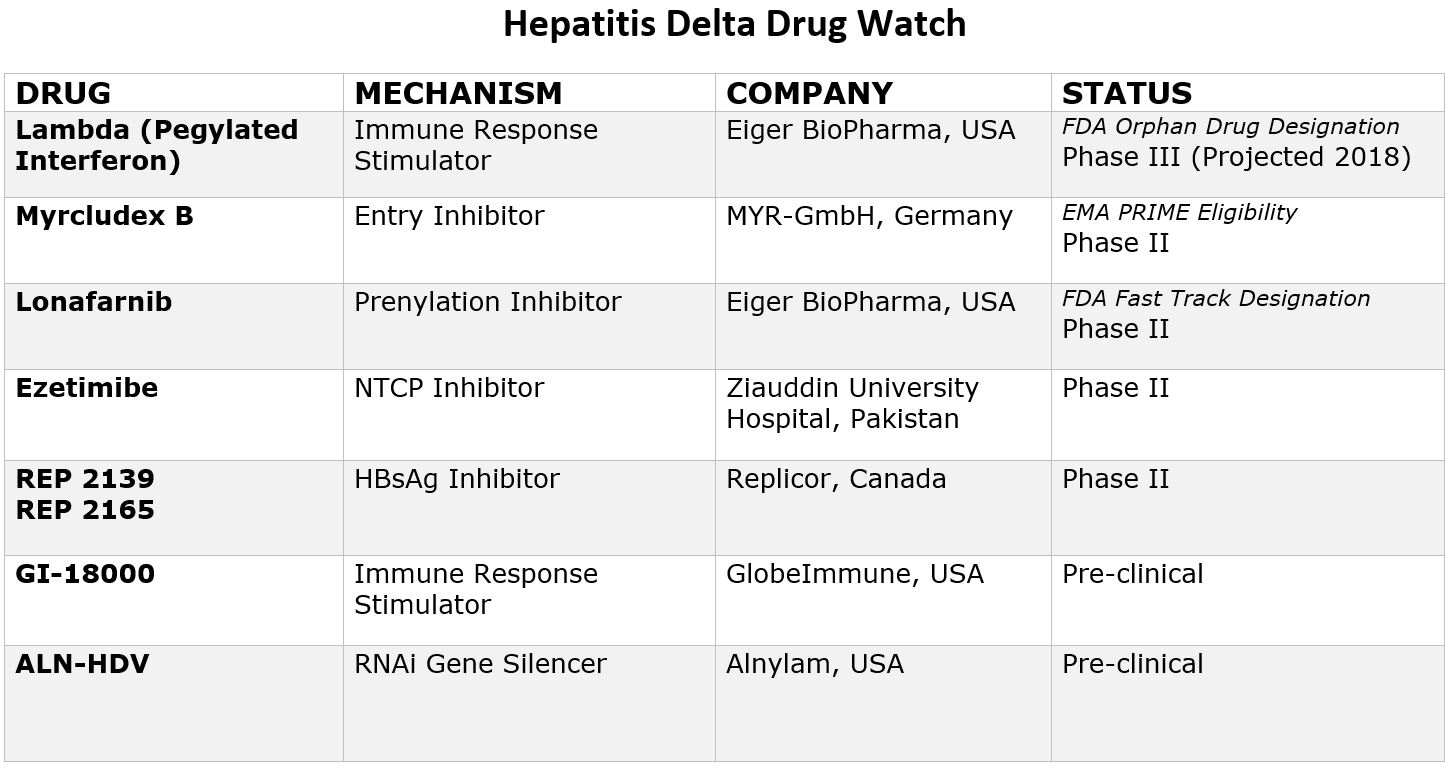
Testing hepatitis B patients for hepatitis delta is important because when people with hepatitis B are exposed to the hepatitis delta virus, 90% will develop a chronic infection1. Coinfection will alter treatment and management plans, because antivirals effective on hepatitis B do not control hepatitis delta2. While the standard treatment of interferon is less than 30% effective in controlling coinfection, there are new drugs in development. With two of these drugs set to enter phase 3 clinical trials in 2019, it is more important than ever to identify coinfected patients and connect patients into clinical trials.
Until recently, only the anti-HDV test was widely available in the United States. In February 2019, Quest Diagnostics began offering HDV RNA testing, making it easier for patients and their physicians to access this more detailed level of testing. A complete list of labs offering testing is below.
Tests Offered:
Tests Offered:
Cambridge Biomedical (US, Limited States)
Tests Offered:
Tests Offered:
Tests Offered:
Centers for Disease Control and Prevention (CDC) (US & International)
Tests Offered:
- HDV Antibody Total
- HDV RNA
- Genotyping
Disclaimer: This may not be a comprehensive list of all available labs offering testing.
Please note, if you are a patient in the U.S. and wish to be tested for hepatitis delta, these tests must be ordered through a clinician.
It is very important for coinfected patients to be managed by a liver specialist who is familiar with managing coinfected patients. For assistance in locating a specialist near you, please visit our Physician Directory page. For additional questions, please visit www.hepdconnect.org, email connect@hepdconnect.org, or call our hotline at 215-489-4900.
References:
- Hooks, B., Billings, J., & Herrera, J. (2009). Hepatitis D Virus. Practical Gastroenterology.
2. Farci, P., & Anna Niro, G. (2018). Current and Future Management of Chronic Hepatitis D. Gastroenterology & hepatology, 14(6), 342-35




 By Sierra Pellechio
By Sierra Pellechio